At a Glance
- The path to a Practical Cure is clearer than it was 15 years ago—but the job is not done.
- Research has transitioned from full-body immunosuppression and basic cell replacement to advanced techniques like gene editing and sBCs.
- Immune system modification remains the most explored pathway, accounting for 64% of currently active projects.
- New for-profit investment is accelerating momentum in the field.
- To turn progress into a cure, the most promising pathways must be pursued with greater urgency and focus. There is no time to waste.
March 26, 2026
2026 marks the fifteenth year JDCA has been working to accelerate a Practical Cure for type 1 diabetes. Much has changed, yet much remains the same. This report takes a look at ‘then’ and ‘now,’ reviewing progress made within the context of a T1D Practical Cure and the remaining challenges.
JDCA started operations in 2011. Since then, T1D cure research has advanced rapidly, while some of the same challenges still exist today. One constant throughout has been that the fight for a cure has never faltered as our community's top priority.
This report is a top-level summary review. Many of the topics addressed would merit their own reports, but here, for the purpose of this overview, we prioritize brevity and readability over volume and depth. We hope it serves as a jumping-off point for further questions, discussion, and ultimately informs priority settings throughout the whole ecosystem.
Practical Cure Project Highlights
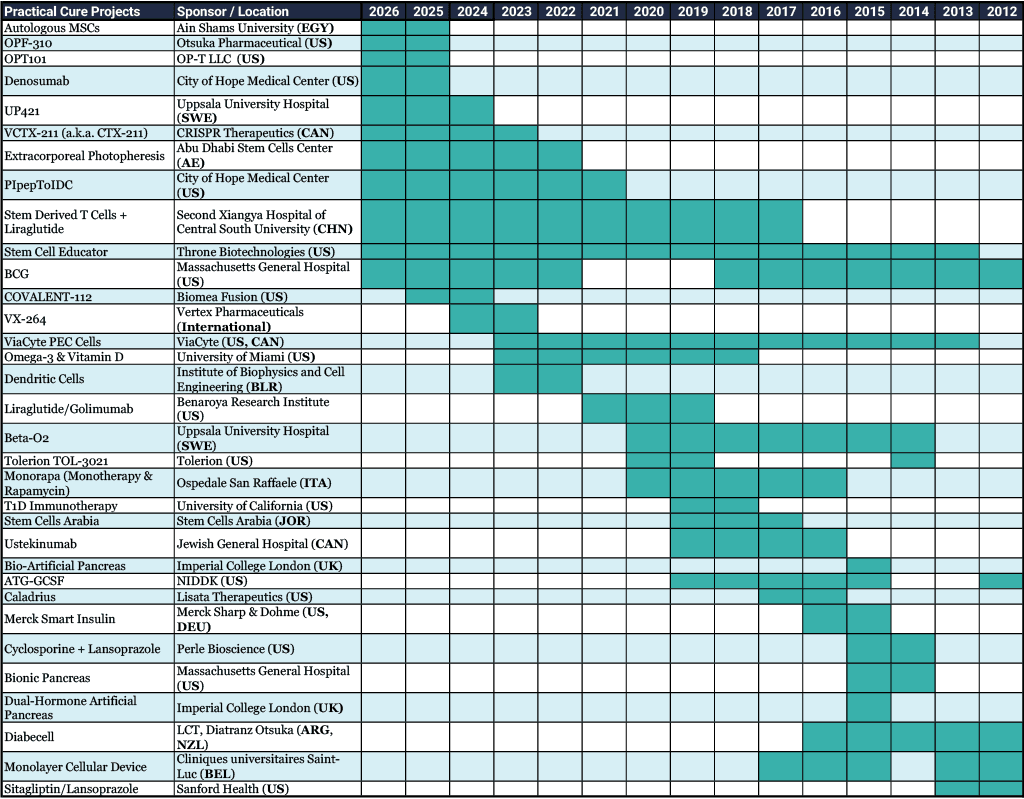
Over the past fifteen years, JDCA has identified and written about thirty-three Practical Cure projects that were, at one time or another, active in human clinical trials. All of these projects were listed in the US clinical trial registry, and several hosted multiple trials testing different patient groups, dosing levels, and other variables (see Appendix A).
Two Projects Remain Active from the Early Years
Most projects undertaken in the early years (2012-2015) did not progress and are no longer pursued today. Only two projects from that period remain in active trials today: BCG and Stem Cell Educator.
A long period of clinical activity is a two-edged sword. While it is good to see research maintain enough positive news and traction to continue development for many years, we would much prefer to see these projects progress up and out of human trials to market approval or, alternatively, allow the resources to be redeployed to more promising research avenues. That said, trial results often tell the story, and we reserve judgment until the next set of results is posted.
Most and Least Explored Cure Research Pathways
The most explored Practical Cure pathway is Immune System Modification, an approach tackling the immune system at its root. 58% of all historical Practical Cure projects sought solutions to modify the immune system as the project’s main or secondary objective. Among the eleven T1D Practical Cure projects active today, 64% address immune system modification.
The least explored pathways are Glucose Responsive Insulin (one trial) and an Advanced Artificial Pancreas (two trials). Neither approach has been on the active Practical Cure list since 2016.
Notably, stem cell-derived beta cells (sBCs), which have attracted so much attention in recent years by publishing promising clinical results, are not the most explored. Only six projects on the clinical trial list were testing a sBC Practical Cure solution. However, today there are a variety of different preclinical sBC solutions that, we hope, will rapidly advance to human trials.
Most Advanced Projects: Stage of Development
None of these projects received market approval, and none have started a full, standalone phase III human trial. Many did not advance beyond phase I, which typically tests safety rather than efficacy. Some trials reached phase II, and others claim they are in the midst of a hybrid PI/PII, or even PII/III.
It is important to note that Vertex is currently conducting a phase III trial for its sBC line. This trial uses broad-spectrum, high-toxicity immunosuppressive drugs to achieve cell protection; therefore, it is not yet a potential Practical Cure and is not included in the list.
Cell Supply and Cell Protection: ‘Then’ vs. ‘Now’
Both key components of a Practical Cure have advanced substantially in the past fifteen years.
Cell Supply
Then: Many Methods, the Need for Sustainable Cell Supply
Methods at the time focused on either regenerating beta cells or directly replacing them via transplantation. The Edmonton Protocol, though ten years old, was still considered a major breakthrough, pushing islet transplantation as a priority research avenue. Xenotransplantation (using non-human animal islet cells) was still being extensively explored. Stem cell-derived beta cells were not a priority research area, even though one aggressive researcher at Harvard, Doug Melton, was making it a focus.
Now: sBCs Are the Story
Today, stem cell-derived beta cells are the primary focus area for sustainable islet cell supply. While there is some continued research on regeneration and xenotransplantation, the clear lead horse is sBCs.
Within the last five years, sBCs have demonstrated efficacy in clinical trials as a sustainable alternative to donor islets, attracting substantial research attention, and the most advanced lines may be as close as a few years away from FDA market approval.
Protection
Then: Full-Body Immunosuppression
At the time, the only way to protect islet cells from the autoimmune attack was with full-body, highly toxic, lifelong immunosuppression. These drugs protected transplanted cells from the immune attack but presented a high risk of serious adverse side effects. The Edmonton protocol included these drugs as an essential component of the procedure. For a small number of people with the most brittle and labile T1D, the risk of these immunosuppressive drugs was considered appropriate, but for most people with T1D, the risks were considered far too great.
Most researchers held the view that there must be an alternative. Encapsulation received substantial attention but faced many challenges. Finding a semi-porous material that offered protection while also allowing them to be responsive to blood glucose was a major hurdle. Issues with fibrosis and ensuring cells received enough blood flow to survive were common.
Now: Advanced Immune Modulating Techniques Being Developed
There is a wide variety of immunomodulating therapies being explored today that incorporate advances in general medical sciences, such as gene editing, monoclonal antibodies, tolerizing vaccines, and others. These capabilities are being directly applied to T1D with the intent to either stop the immune system from attacking beta cells or hide transplanted cells from the autoimmune attack.
A key breakthrough was made last year by Sana Biotechnology and Uppsala University Hospital. In clinical trial UP421, beta cells gene-edited to hide from the immune system without immunosuppression continued to survive and produce insulin, a result that has persisted for 14 months post-procedure.
Although only one patient was dosed—enough for excitement but not enough for certainty—this research success is attracting a number of other for-profit companies and academic researchers to undertake projects that render sBCs invisible to the immune system.
Funding & Research
In 2011, nonprofits and government agencies were the main funders of T1D cure research, which was primarily conducted at academic research centers and universities. The approach was broad, with funding spread across many research pathways and projects. At the time, there did not appear to be a clear lead horse approach to solving cell supply, nor one for solving cell protection. There was some for-profit exploration, but only in small numbers.
Today, we stand at a pivotal juncture. Some curative research pathways are increasingly led and funded by companies, large and small, as well as by nonprofit and government grants. This is a major change that could drive substantially more research dollars toward T1D.
The root cause of the shift is that T1D research pathways have become more attractive to for-profit companies. The successful research results in clinical trials for sBCs and engineered cells are attracting for-profit companies seeking to capitalize on these developments. In 2016, Breakthrough T1D founded the T1D Fund, an independently operated subsidiary that uses venture philanthropy investment techniques to fund T1D research led by for-profit entities, marking a significant turning point in generating for-profit interest.
Nonprofit and government grants will still be essential, but they will play a much more focused role. Specifically, their optimal roles are to unlock the most complex and stubborn research problems and prepare the marketplace so that any future treatment is available for everyone with T1D as quickly as possible.
What Has Not Changed
Despite the many changes addressed earlier in this report, we must not take our foot off the gas. The need for ‘More for a T1D Cure’ remains today as important as ever.
For many years, JDCA argued that a paradigm shift was needed to focus intensely on the most promising research pathways. This shift is as necessary today as it was in 2011.
While we are moving in that direction with lead-horse approaches in cell supply and cell protection, we must ensure that the most promising research pathways are fully funded so they can be developed, tested, and achieve results—whether successful or not—as quickly as possible.
The T1D research ecosystem remains tuned to spreading its resources across many areas of research rather than doubling down on the most promising ones. Together, we must resist this and encourage—perhaps demand, for the sake of everyone battling T1D—a deeper, sharper, more urgent focus on the most promising research. There is no time to waste.
Appendix A: Overview of Historical Practical Cure Projects (2012-2026)