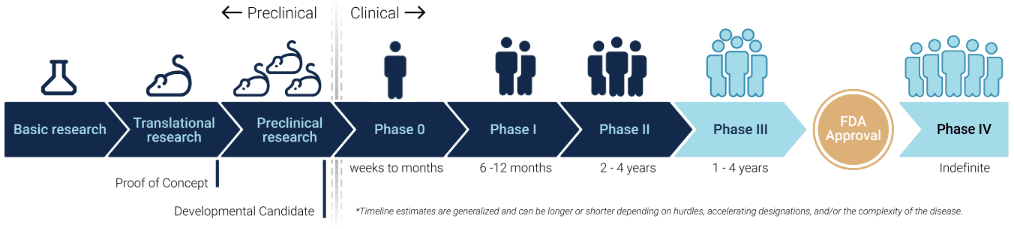
- A therapy moving through clinical trial phases can take 10-15 years to before hitting the market.
- Most research begins by testing basic safety in phase I and ends by proving efficacy and safety in large phase III trials, the last step before market approval.
- Phase IV trials are sometimes required after market approval to test ongoing safety in very large, real-world patient populations.
- The use of ‘seamless’ trials like I/II or II/III (where trials flow seamlessly from one phase to the next) or specialized FDA designations can expedite clinical trial timelines.
April 16, 2026
This report aims to provide an easy-to-understand reference of how medical research moves from early-stage animal research through each stage of human trials. Every approved therapy in the market today has gone through this process. The journey through all human clinical trial phases can take ten to fifteen years if all goes well, and, as research progresses through each stage, the hurdles steepen, and costs rise sharply. Understanding what happens in each phase of development is key to understanding the T1D research landscape.
This report will illuminate the purpose, structure, and typical timelines associated with each clinical trial phase. There is also some confusion when companies label their trials in compound terms, like phase I/II or II/III. This report will untangle and explain these and other key aspects of human trials.
Before Human Trials Begin
A massive amount of development work occurs before the first human clinical trial begins. The research must progress from a hypothesis to laboratory validation and into animal testing. Most often, many years are spent in preclinical development. Only when there is sufficient evidence that the research pathway is both safe and effective, is it considered for human trials.
At that point, the first step to human trials is to file an Investigational New Drug (IND) application with the FDA. The application must demonstrate sufficient safety and efficacy from earlier testing, and lay out the framework for the first trial.
IND approval is not an endorsement of efficacy by the FDA. Instead, it simply means the therapy and trial design are safe and appropriate, and there is enough therapeutic potential to justify further evaluation.
Human Clinical Trial Phases
With an IND in place, researchers can begin human trials. Each clinical trial phase is a stepping stone, gathering the data necessary to progress safely to the next tier.
There are five phases of human trials, from Phase 0 through Phase IV. Phases I, II, and III are mandatory. Phase 0 is an optional early exploration trial that some researchers conduct before committing resources to a full Phase I. Phase IV commences after the FDA grants market approval for products that merit heightened, ongoing surveillance in the broader population.
The phases are described in more detail below.
Phase 0: Preliminary Testing (Optional)
Phase 0 is an optional, somewhat rare step before initiating full phase I, designed to gauge early safety. Here, a small group of individuals is given a very small microdose of the intended therapy to see how the body tolerates the drug before entering into a longer and more expensive phase I.
- Purpose: To gather preliminary data on safety, toxicity, and the body’s handling of the drug before phase I, cutting significant costs associated with further trials if the drug is unsafe. No therapeutic effect is expected.
- Patient Size: 10-20, healthy volunteers and/or patients with the disease.
- Dosing: Microdose to all patients, typically 1/100th of the target dose.
- Time: A few weeks to months.
Phase I: Safety (Mandatory)
Phase I is the step where most ‘first-in-human’ clinical trials begin, determining a new drug’s safety. The primary objective is to test how the body tolerates the drug and to find the best dosage.
Early data here is critical, deciding future patient eligibility, endpoint criteria, influencing future funding opportunities, and most importantly, whether the therapy is safe enough to continue.
- Purpose: To test safety, determine the appropriate dose, and see if future trials are warranted.
- Dosing: Either (1) using a single ascending dose that rises over time or (2) multiple ascending doses where different cohorts (groups) receive different amounts.
- Patient Size: 20-100 healthy volunteers (primarily) or patients with the disease.
- Time: General estimates range from 6 to 12 months. Trials may take several years if the drug or disease is complex.
- Success: About 70% of drugs generally progress to the next phase.
Phase II: Efficacy (Mandatory)
Phase II beings seriously asking the question, “Does it work?” in addition to monitoring for any adverse events. This stage must collect enough data to warrant a phase III trial.
- Purpose: To gauge true efficacy in the intended population while monitoring adverse effects.
- Patient Size: 100-300 patients with the disease.
- Dosing: The full therapeutic dose. Different cohorts test the drug in different populations or include a placebo group (single blind). Some trials are double-blind, meaning neither researchers nor patients know who received the drug until data is collected.
- Time: 2-4 years. Trials may take several years if the drug or disease is complex.
- Success Rate: About 33% of drugs generally progress to the next phase.
Phase III: Proof in a Larger Population (Mandatory)
This is the last phase before hitting the market, the turning point between FDA approval or failure. Here, a drug needs to demonstrate efficacy in as many patients as possible while avoiding adverse effects that may not have appeared in smaller trials. Notably, this phase is estimated to account for 60% of total drug development costs, putting a lot on the line if results don’t perform.
- Purpose: To determine if the therapy is safe and effective on a large scale.
- Patient Size: 300-3000 patients with the disease. Patients could have other diseases and be on other medications that could impact trial data.
- Dosing: The full therapeutic dose is given. Often, trials are double or single blind.
- Time: 1-4 years. Trials may take several years if the drug or disease is complex.
- Success Rate: 25-30%.
FDA Pre-Market Approval
After a successful phase III, the principal investigator may seek FDA approval to bring the new drug or medical product to market. They will submit either a New Drug Application (NDA) or a Biologics License Application (BLA) to the FDA for review. FDA scientists review the applications, including a summary of all testing data, and make an informed recommendation to the panel of decision-makers. Ultimately, it is the job of the FDA to keep Americans safe and to ensure that benefits outweigh any outstanding risks.
During this process, the FDA also reviews the plans for marketing, manufacturing, and ongoing quality control. They ensure that the proposed product labeling is accurate and that any manufacturing plan can maintain consistent quality, identity, purity, and strength. Lastly, if there are any ongoing unique concerns with the product, they will establish a protocol of surveillance and reporting that will continue even after the product is on the market.
The approval process as a whole can take upwards of a year. Overall, only 8.5% of therapies that enter phase I clinical trials are approved for the market.
Phase IV: Post-Market Surveillance (Often Required)
Phase IV is the last clinical trial phase and occurs after market approval. If needed, this phase is used to monitor long-term side effects in the broader population. The specific surveillance required in phase IV is defined during the Market Approval process.
Most therapies based on newer, more complex medical science will require phase IV ongoing trials, which will likely include most of the research for a T1D Practical Cure. Stem cell-derived beta cells, which may solve the cell supply issue, are a relatively recent medical breakthrough of which we are still learning. Any treatment that involves altering the immune system is complex and presents opportunities for long-term side effects. We expect that any functional cure that receives FDA approval will include a robust ongoing study protocol, which is a good thing.
Phase IV is an extra level of insurance and protection for the consumer and should not be confused with the normal risk surveillance the FDA manages for all pharmaceuticals. If new data emerges regarding serious adverse effects or long-term health concerns, a drug may be pulled from the market or limited to specific patient populations.
- Purpose: To gauge safety over an extended period of time in real-world, large patient populations.
- Patient Size: Hundreds to thousands of patients.
- Dosing: The study is observational, monitoring patients who already take the approved drug.
- Time: Indefinite. Studies can range from a few years to several decades.
Additional Information
There are two additional topics that are important in understanding human trials. Both offer the opportunity to accelerate testing speed.
1. Combined Phases: Seamless Trials
Today, it is not unusual to see trials labeled as I/II or II/III, which is called a 'seamless' trial. Seamless trials are designed to allow for one phase of testing to flow seamlessly into the next without stopping. The trial label is chosen by the lead investigators, not the FDA. The FDA gives the investigator wide discretion to label the trial, but ultimately requires the same quality of data whether the trial is a seamless or stand-alone phase.
Over half of the projects on our Practical Cure list use these labels.
The benefit of using a combined phase trial is to transition smoothly from one phase to another, without stopping to reevaluate the protocol or submit a new study application. This ensures data is continuously gathered and can reduce costs. Before a seamless trial can begin, the trial objectives, enrollment, and other critical factors must be clearly laid out.
The drawback is that the complexities of a seamless trial complicate the analysis of trial data. Additionally, it can be tricky to tell if a project is actually operating in the earlier or later phase.
2. Expediting US Clinical Trial Timelines
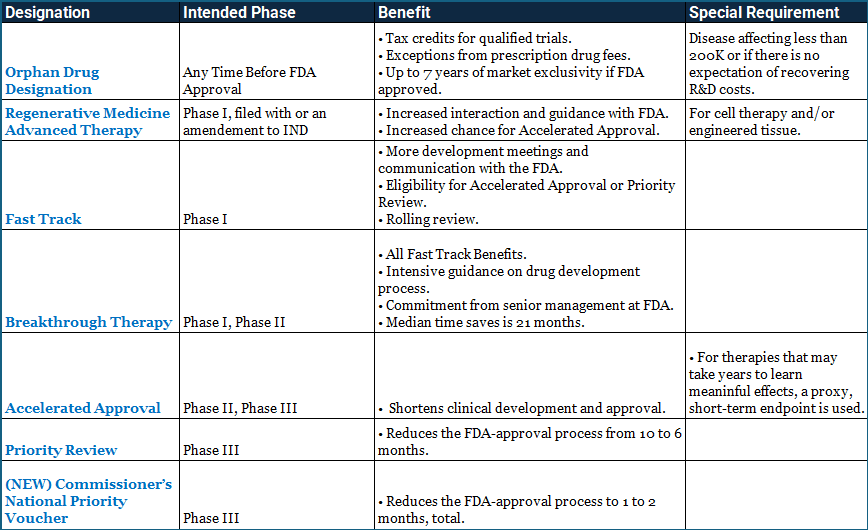
There are seven designations that the FDA may provide to accelerate the approval process for research progressing through human trials.
All of the ‘fast-track’ designations below are contingent on the therapy addressing an unmet need for a serious condition, being superior to existing therapies (e.g., reducing side effects), and/or greatly improving patient outcomes in the cure, treatment, or prevention of a disease. Please note that granting a designation does not endorse the therapy’s efficacy.
Therapies in clinical trials can receive more than one designation. It’s estimated that being granted one designation shortens the median time to first approval for drugs by fifteen months.