Last Thursday and Friday, the University of Toronto hosted a virtual research conference that featured some of the world’s most prominent diabetes investigators. The conference, called “Insulin 100,” commemorated the 100th anniversary of the discovery of insulin.
This report summarizes the essential points discussed at the conference related to a type 1 diabetes cure.
Conference Overview
In the spirit of the discovery of insulin at the University of Toronto 100 years ago, this virtual conference aimed to “provide comprehensive updates on the latest advances in diabetes treatment and management.” The conference agenda covered a wide range of topics from cutting-edge research to patient care. All sessions were recorded and are available for free on the event’s website and can be accessed by clicking the button at the bottom of this email.
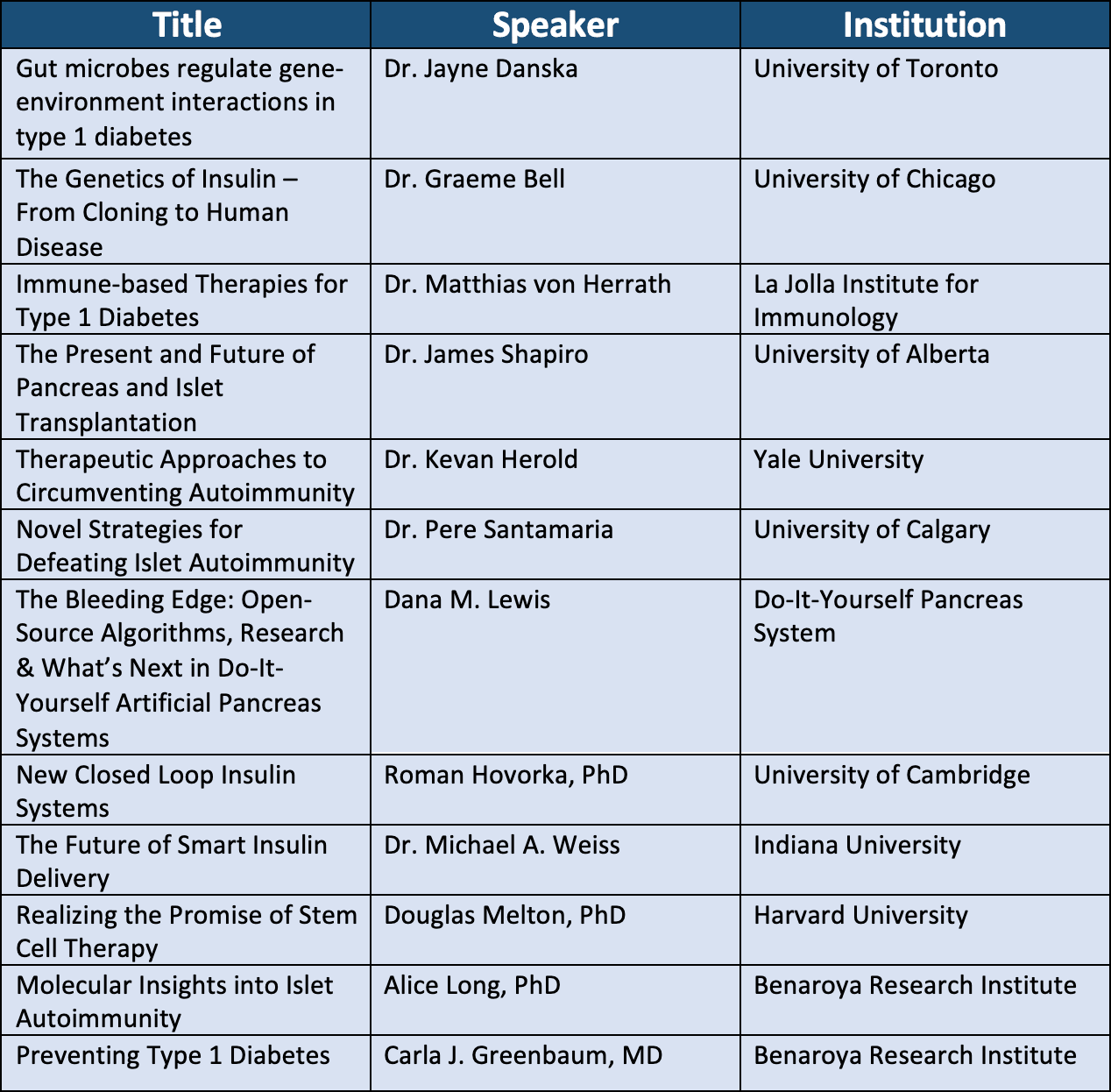
In total, there were 37 pre-recorded presentations which each corresponded with a live “Q&A” session, 12 of which addressed topics that relate to a T1D Practical Cure. A list of the 12 relevant presentations is included in the appendix.
T1D Cure Takeaways
- Among many esteemed speakers, the event offered the rare opportunity to engage in Q&A sessions with both Drs. James Shapiro and Doug Melton, pioneers in the field of islet and beta cell replacement therapy.
- Dr. Shapiro announced that his lab hopes to start a human trial investigating stem-derived beta cells supplied from the patient's blood cells later this year. This is an exciting advance addressing the need to increase the availability of beta cells for implantation.
- Dr. Melton expressed a bullish outlook on gene editing. He shared two possible avenues where this emerging capability could improve beta-cell therapy: 1) by increasing the efficacy of transplanted cells and 2) by protecting implanted cells from the T1D autoimmune attack.
- Dr. Carla Greenbaum, another T1D research luminary, listed seven immune modifying drugs which have been shown to alter the progression of T1D in people recently diagnosed with the disease.
- Dr. Greenbaum is the primary investigator in a trial testing the efficacy of one of those drugs – Golimumab – in people with fully established T1D. The JDCA has noted this therapy as a potential Practical Cure for T1D; Dr. Greenbaum did not share any updates on the trial.
- Additional highlights included an update of Beta Bionics’ dual hormone artificial pancreas system and a presentation about the status of Glucose Responsive Insulin therapies.
- Dr. Steven Russell of Beta Bionics detailed the commercialization timeline for the company’s dual hormone (insulin + glucagon) pump system. Beta Bionics’ “iLet” is projected to start human trials at the end of this year, with anticipated FDA approval in 2023.
- Dr. Michael Weiss, who co-founded Thermalin, Inc. – a company exploring the potential of glucose-responsive insulin (GRI) – gave an overview of the current research landscape. GRI remains an exploratory area of study with no current active human trials.
Appendix: Discussion Sessions Related to a T1D Practical Cure