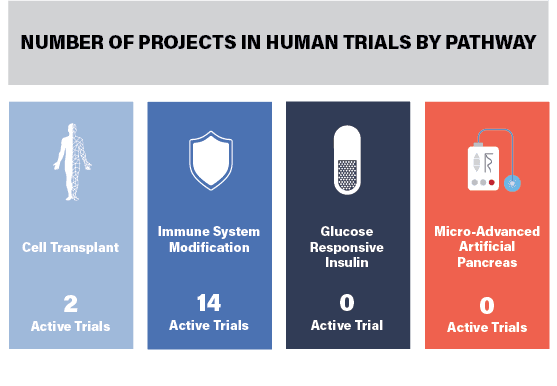
This report reviews the 11 active Practical Cure (PC) research projects underway in FDA approved human clinical trials. The 11 projects are conducting a total of 16 trials, as illustrated in the opening chart and noted in the trial summary below along with the project title, description, location, and status. A full definition of a Practical Cure, as well as the four research pathways, can be found in last week's JDCA report, The Four Research Pathways to a Practical Cure for Type 1 Diabetes (Click here to view). No projects have been abandoned since our last review in the 2018 State of the Cure and two new projects have been added to the list.
Projects by pathways are listed below.


Definition:
- Therapy to stop the immune system from destroying beta cells, including modifying, blocking, and retraining.

Definition:
- Implanting islet cells, stem cells, or precursor cells to achieve insulin independence. Cells are protected by an encapsulation device or immune system modification.

Definitions:
Glucose-Responsive Insulin: Insulin which chemically activates in response to changes in blood sugar.
Micro-Advanced Artificial Pancreas: A device that mimics the pancreas by monitoring changes in blood sugar and independently administers insulin without the patient’s input. To be considered a Practical Cure the device must be small enough for users to completely forget about its presence. No artificial pancreas projects in, or nearing, human trials are currently small enough.
